Real World Evidence
A CDSCO & ISO-Certified Partner for RWE Studies
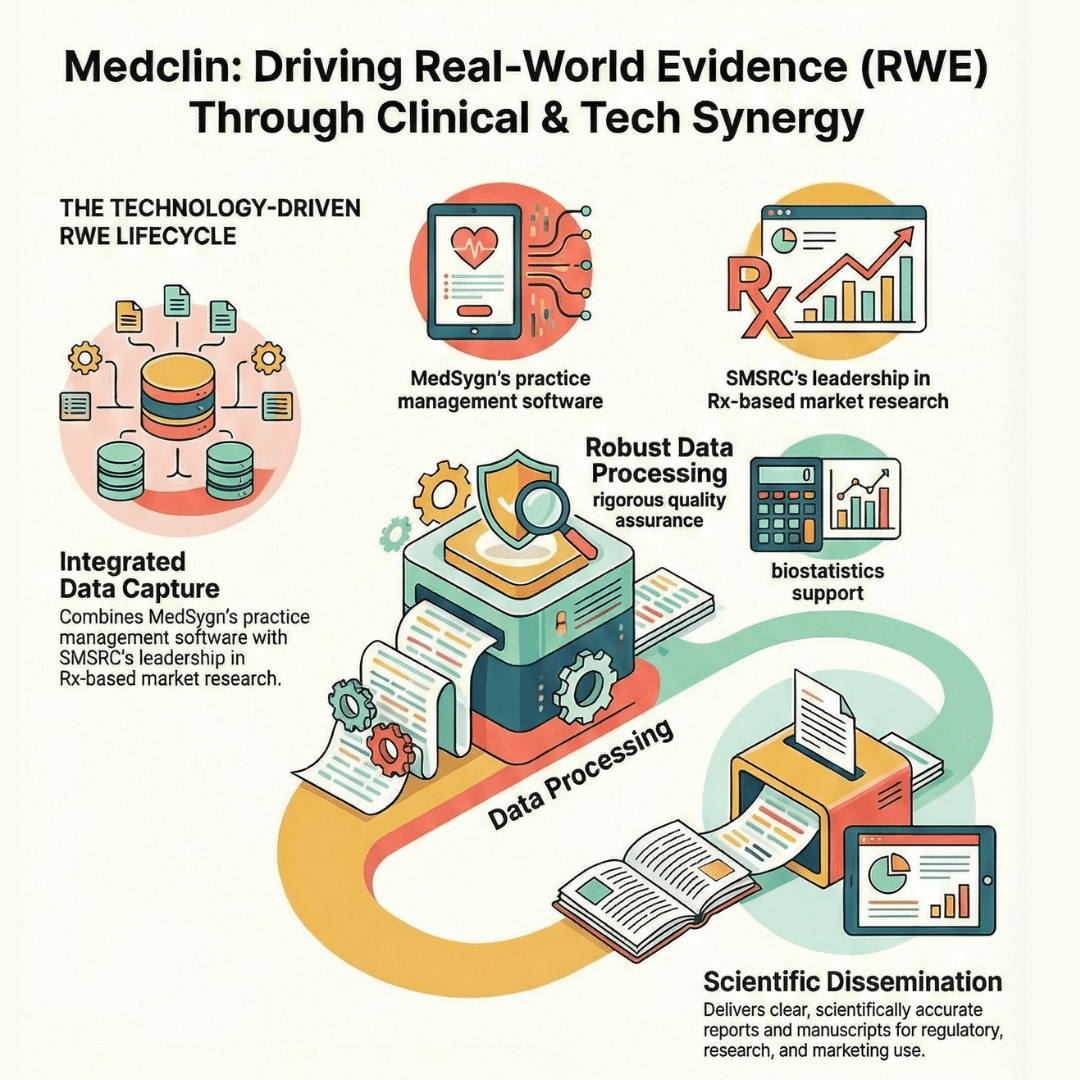
Medclin conducts Real-World Evidence studies that bridge real-world clinical practice with scientific evidence.
Our approach combines large-scale real-world data, longitudinal patient follow-ups, and robust analytics to evaluate effectiveness and safety.
The resulting evidence supports regulatory, medical, and strategic decision-making across product lifecycles.
- End-to-End RWE Study
- CDSCO and ISO 9001:2015 certified
- Deep Industry Integration
Compliant, and execution-focused RWE capabilities
Clinical leadership driving real-world insights
RWE studies are conceptualized and guided by senior physicians, epidemiologists, and medical affairs experts who translate routine clinical practice into structured, publishable real-world evidence.
RWE study design anchored in real clinical practice
Comprehensive management across pre-study, study, and post-study phases — from protocol development and site initiation to database lock, clinical study reports, and publication in peer-reviewed journals.
Regulatory, ethics & quality compliance
Phase studies conducted under CDSCO-aligned and ISO 9001:2015 certified operations, supported by a robust Quality Management System aligned with ICH-GCP and regulatory requirements.
Large-scale real-world data access & processing
RWE studies leverage structured real-world data from electronic medical records, physical prescriptions, and integrated Rx databases—processed through secure, validated, and scalable data systems.
Integrated phase study services
Medclin conducts both longitudinal and cross-sectional RWE studies, enabling analysis of treatment effectiveness, safety, patient-reported outcomes, and real-world clinical decision-making over time.
Patient-reported outcomes and real-world follow-ups
Flexible phase study design and execution tailored to protocol requirements, therapeutic area needs, and sponsor objectives, supported by strong site intelligence and investigator networks.
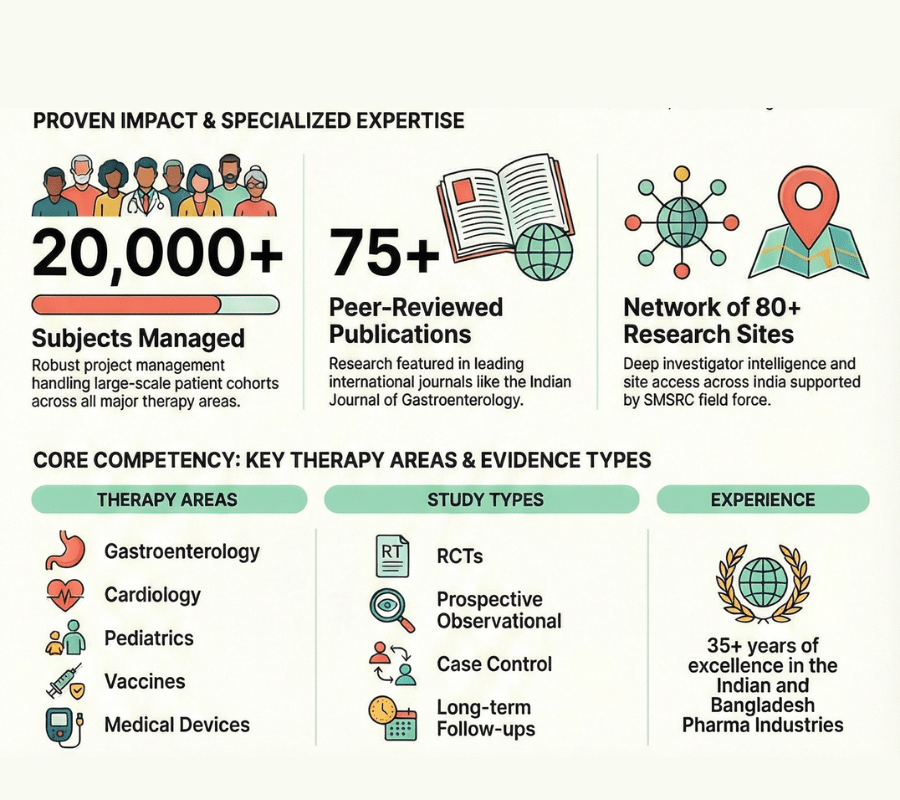
20,000 Projects, 75 Publications
Trusted by Pharma Giants












© 2026 All Rights Reserved.
