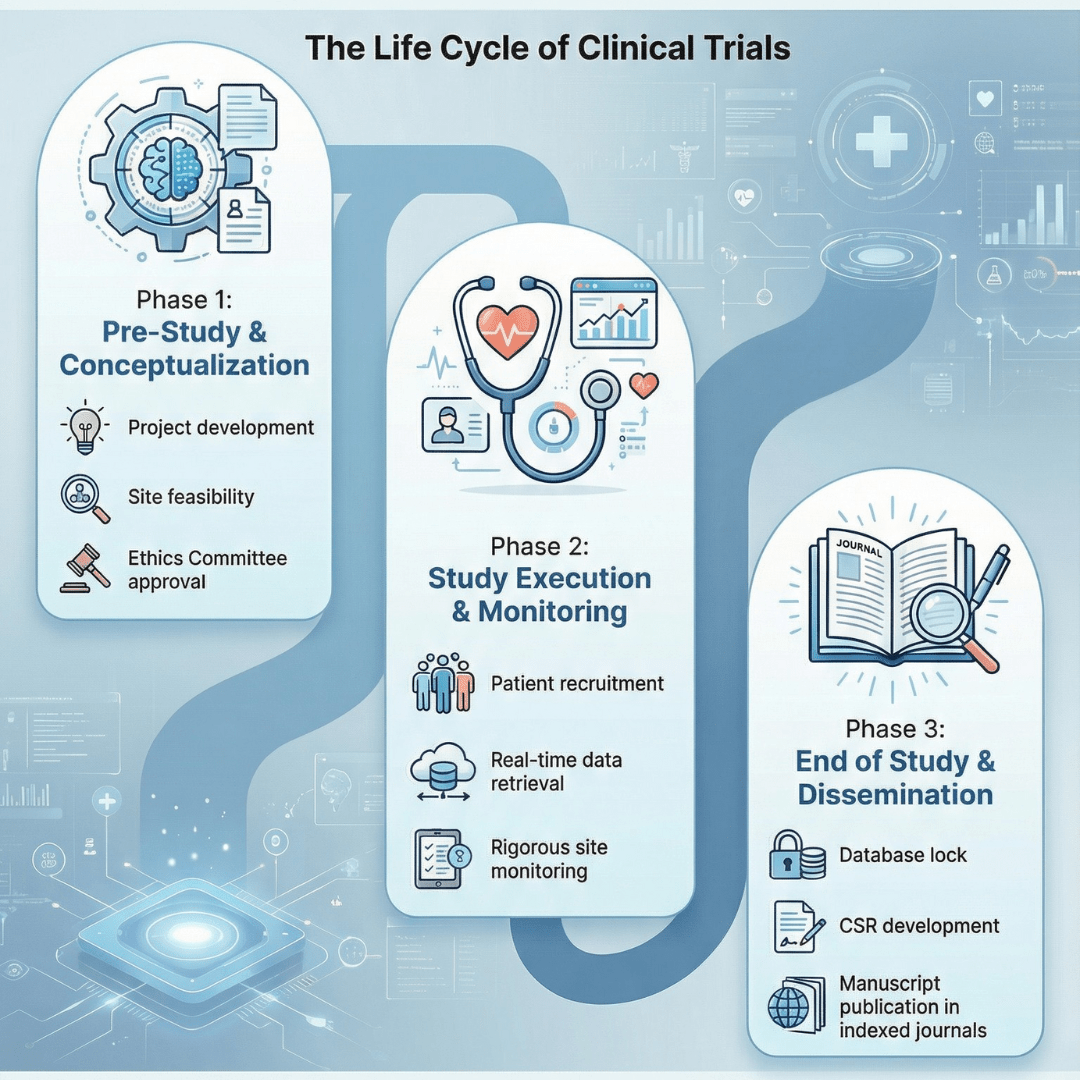
Phase Studies
A CDSCO & ISO-Certified Partner for Phase Studies
Medclin Research conducts phase studies in close collaboration with medical professionals, investigators, and the pharmaceutical industry. Our approach integrates strong medical leadership, academic expertise, and key opinion leader (KOL) involvement to design scientifically sound study protocols and generate reliable clinical evidence that supports informed healthcare decision-making.
- End-to-End Phase Study Services
- CDSCO and ISO 9001:2015 certified
- Deep Industry Integration
Compliant, and execution-focused Phase Study capabilities
Strong medical leadership for phase studies
Phase studies are led by senior physicians, academicians, and principal investigators with decades of experience across IPM, pediatrics, vaccines, and multiple therapeutic areas, ensuring scientifically sound study conceptualization and execution.
End-to-end phase study lifecycle management
Medclin designs RWE studies based on real prescribing behavior, treatment pathways, and patient outcomes—ensuring research questions are relevant to clinicians, regulators, and market access teams.
Regulatory-aligned and publication-ready RWE execution
All RWE studies are executed under CDSCO-aligned and ISO 9001:2015 certified quality systems, ensuring data integrity, audit readiness, and acceptability for regulatory and scientific dissemination.
Secure, accurate & audit-ready data management
Dedicated data management teams ensure validated, encrypted, and high-quality datasets with complete documentation, supporting regulatory submissions and audit readiness throughout the study lifecycle.
Longitudinal and cross-sectional RWE capabilities
Phase studies supported by integrated capabilities including site management, monitoring, biostatistics, medical writing, and quality assurance — enabling seamless coordination across all study functions.
Study-specific & sponsor-centric execution
RWE studies incorporate patient follow-ups, outcome reporting, and experience-based data collection through structured monitoring and digital engagement mechanisms.
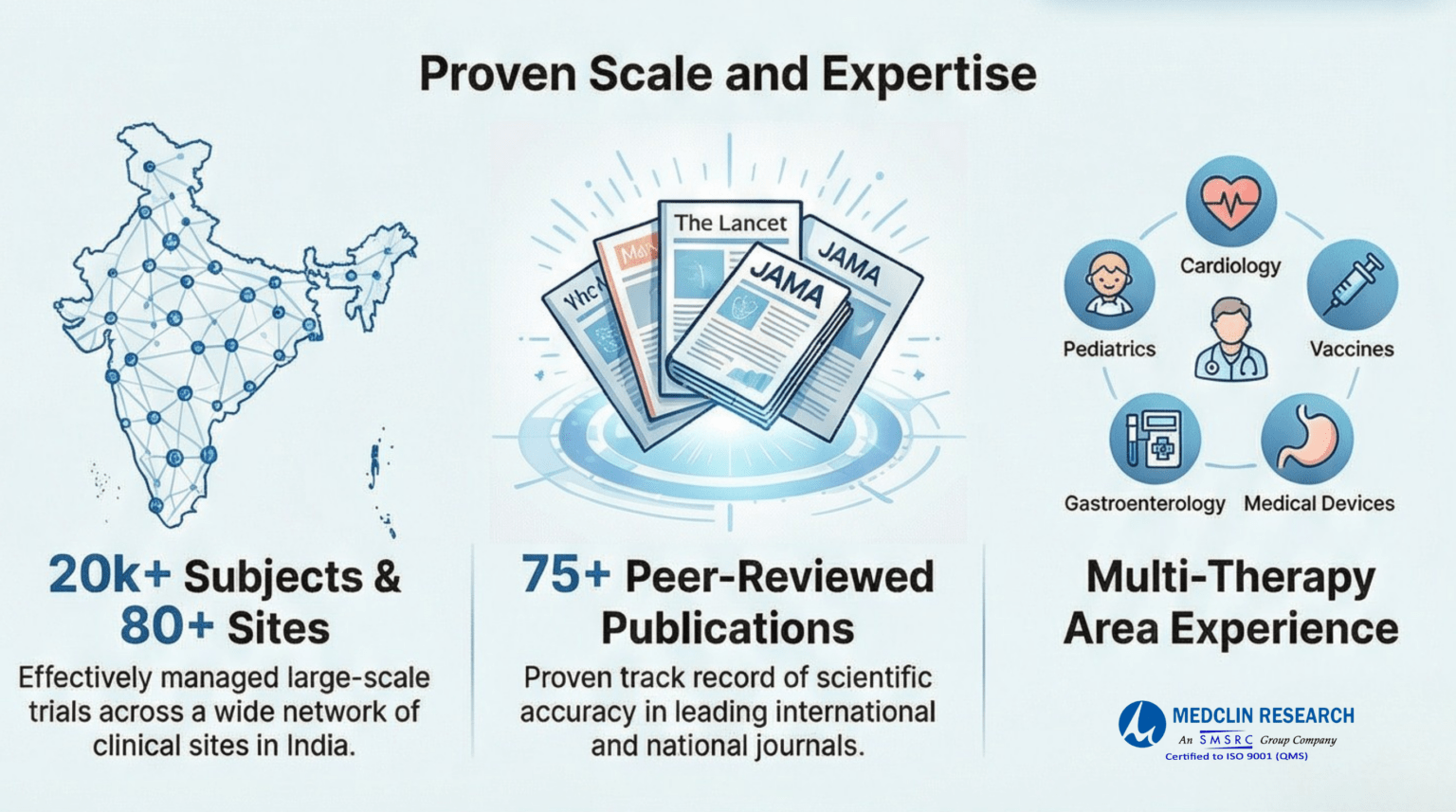
20,000 Projects, 75 Publications
Trusted by Pharma Giants












© 2026 All Rights Reserved.
