Phase Studies
Phase II–IV Clinical Trial Management
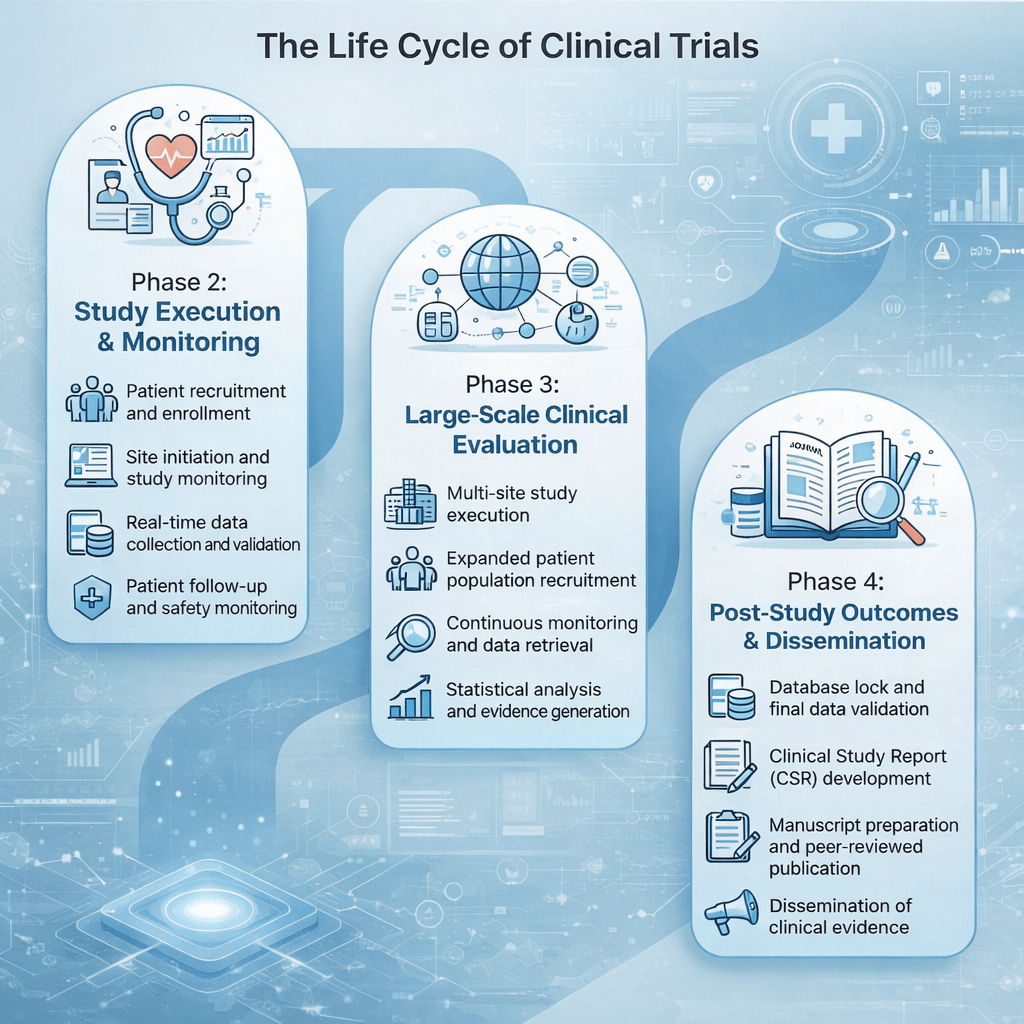
Medclin conducts late-phase interventional studies and post-marketing research across multiple therapeutic areas, supporting sponsors in evaluating safety, effectiveness, treatment patterns, and patient outcomes in real clinical settings. |
- Study feasibility and site identification
- Regulatory and ethics committee submissions & coordination
- Investigator and site management
- Patient recruitment and retention support
- Clinical monitoring and project management
- Data management and quality oversight
- Medical review and clinical study reporting
With strong investigator networks and operational experience, we focus on efficient study execution, regulatory compliance, and high-quality data generation aligned with ICH-GCP standards.
Late-phase trials are critical for understanding real-world effectiveness and optimizing treatment strategies, and CRO partners play a key role in ensuring timely and compliant study delivery.
Compliant, and execution-focused Phase Study capabilities
Strong medical leadership for phase studies
Phase studies are led by senior physicians, academicians, and principal investigators with decades of experience across IPM, pediatrics, vaccines, and multiple therapeutic areas, ensuring scientifically sound study conceptualization and execution.
End-to-end phase study lifecycle management
Medclin designs RWE studies based on real prescribing behavior, treatment pathways, and patient outcomes—ensuring research questions are relevant to clinicians, regulators, and market access teams.
Regulatory-aligned and publication-ready RWE execution
All RWE studies are executed under CDSCO-aligned and ISO 9001:2015 certified quality systems, ensuring data integrity, audit readiness, and acceptability for regulatory and scientific dissemination.
Secure, accurate & audit-ready data management
Dedicated data management teams ensure validated, encrypted, and high-quality datasets with complete documentation, supporting regulatory submissions and audit readiness throughout the study lifecycle.
Longitudinal and cross-sectional RWE capabilities
Phase studies supported by integrated capabilities including site management, monitoring, biostatistics, medical writing, and quality assurance — enabling seamless coordination across all study functions.
Study-specific & sponsor-centric execution
RWE studies incorporate patient follow-ups, outcome reporting, and experience-based data collection through structured monitoring and digital engagement mechanisms.
© 2026 All Rights Reserved.
